Efficacy of Xeljanz, Biologics, & DMARDs in Rheumatoid Disease
Why should I feel like I failed? I’m not alone
 We probably agree it’s unfair to say, “patients fail treatments” instead of the reverse, but there is still disappointment. I was optimistic that my Rheumatoid disease would respond to Xeljanz, the new oral DMARD (disease modifying anti-rheumatic drug) from Pfizer. Unfortunately, my joint stiffness and inflammation, hoarseness, and fevers continued. However, the vascular rash on my leg (diagnosed as livedo reticularis) improved while taking Xeljanz, and it worsened after I stopped taking it.
We probably agree it’s unfair to say, “patients fail treatments” instead of the reverse, but there is still disappointment. I was optimistic that my Rheumatoid disease would respond to Xeljanz, the new oral DMARD (disease modifying anti-rheumatic drug) from Pfizer. Unfortunately, my joint stiffness and inflammation, hoarseness, and fevers continued. However, the vascular rash on my leg (diagnosed as livedo reticularis) improved while taking Xeljanz, and it worsened after I stopped taking it.
Should any of this help you decide whether to try Xeljanz? Probably not. The data shows:
- a difference exists between placebo and tofacitinib (Xeljanz);
- a wide variety of response exists between patients; and
- a small minority respond really well.
A minority has an excellent response to Xeljanz and Biologics
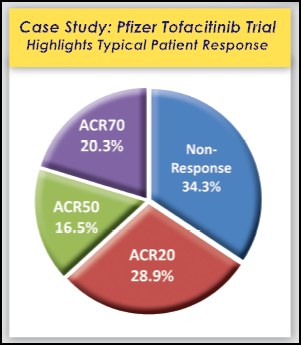
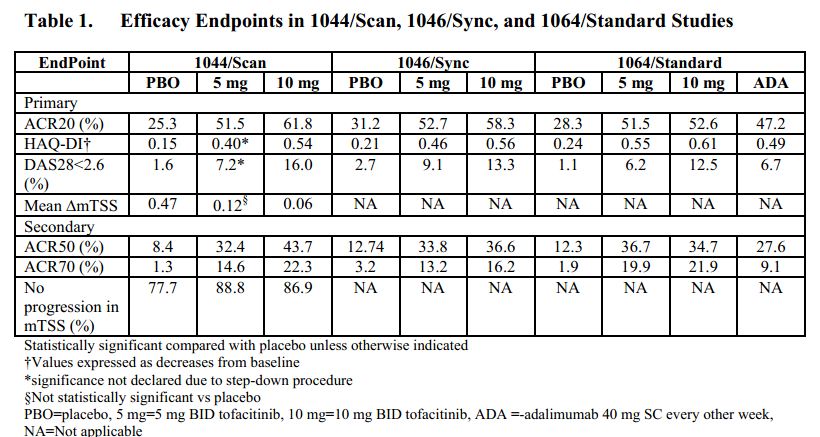
The efficacy data with Xeljanz differ among their various trials, but they are similar. And response levels are comparable to those of Biologics, using ACR20, ACR50, and ACR70. Three years ago on RAW, we discussed some comparisons of efficacy of Biologics, according to a Cochrane review of 31 studies of ACR50 and Danish study of ACR70. Looking at trial data and comparison studies, on average, about 34% are non-responders, which means they do not meet the primary endpoint of ACR20 (an assessment of 20% improvement according to specific measures of disease activity). The remaining 66%, considered responders, are divided between ACR20, ACR50, and ACR70. On average, 20% or fewer people experience 70 percent improvement, and about 29% reach ACR20 only. The remainder are ACR50 only.
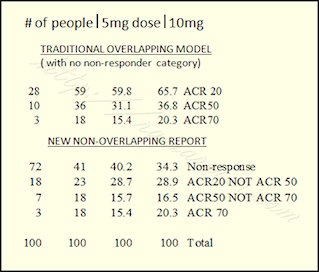 Remember, the ACR response levels are usually reported with all higher responses included, so the traditional ACR20 number includes all responders (including ACR50 and ACR70). It may be easier to understand showing all groups separated in a traditional pie chart (see images to right and at top). That method is discussed here on RAW: New Way to Report Response in RA Clinical Trials?
Remember, the ACR response levels are usually reported with all higher responses included, so the traditional ACR20 number includes all responders (including ACR50 and ACR70). It may be easier to understand showing all groups separated in a traditional pie chart (see images to right and at top). That method is discussed here on RAW: New Way to Report Response in RA Clinical Trials?
Are Biologics and Xeljanz more effective than traditional DMARDs like methotrexate?
Sometimes people assume that shots are stronger than pills, and IV infusions are stronger than shots. But that’s not accurate; the medications work by different mechanisms, so they are effective on different patients with different disease mechanisms and progression of disease. It’s true that Biologics and Xeljanz are considered more effective than other DMARDs, but that could be misleading in some ways: To people who respond well to methotrexate alone, or even Plaquenil, DMARDs are sufficiently effective.
The complexity of Rheumatoid disease, its heterogeneity, and tendency to change in the same patient over time all complicate measurement of efficacy, as well as development of new treatments.
That said, traditional DMARDs are considered less effective, and this has been shown in head to head studies. As a matter of fact, one thing companies do during trials is attempt to show their new product is more effective than methotrexate. As RAW reported last year, Xeljanz was found superior to methotrexate. A couple of years ago, RAW reported on testing of a new possible methotrexate alternative and a British Medical Journal review of methotrexate studies showing 46% reached ACR20 and only 23% reached ACR50 (by the inclusive method).

Some trials have also compared combinations of DMARDs together (triple therapy) or combinations of a DMARD and a Biologic. So far, it has often been concluded that methotrexate with a Biologic is best at preventing radiologic damage (joint damage as studied on x-ray).
Bottom line for Rheumatoid patients choosing therapies
The bottom line is that some respond very well to currently available therapies, but the range of responses is wide, and there is no way to predict yet which treatment or combination will work best. As we discussed recently on RAW, people often weigh risks and benefits for choices, including medications. It has been my own personal choice to try every opportunity available to increase function and get relief from pain, and I’ve made that choice with no regrets.
Recommended reading
- Xeljanz Rejected by European Medicines Agency
- Which Biologic Treatment Should RA Patients Try Next?
- Comparison of Biologics for Rheumatoid Arthritis (RA)
- A Methotrexate Alternative for Rheumatoid Arthritis?
- Rheumatoid Arthritis Drug Approved by US FDA: Pfizer’s Xeljanz Pill (Tofacitinib)
- Xeljanz Cost with a Copay Card, Free Samples, Efficacy, & Side Effects: Gathering Rheumatoid Arthritis Patient Stories
- All articles on Biologics on RAW




rawarrior I’m glad to hear that your vasculitis improved & send regrets you didn’t get a much improved #Rheum response with #Zeljanz.
Unfortunately I did not respond to Xeljanz either. Over 3 months of taking it my pain, stiffness and fatigue increased substantially. As the majority of my pain is in my feet my rheumatologist has now recommended Actemra and I had my first infusion last Friday. Fingers (and everything else) crossed….
good luck Lisa. I hope it helps.
Hi Lisa
Hows it going with the switch back to Actmera.. I having same issue went on Xeljanz now for 3 months and well have pain in the neck and feet and feel tired etc
I think the Actemra was better was on it for a year..
Rgds
Rory
Started Xeljanz recently myself. After a couple of weeks, I developed the “obligatory” upper respiratory infection…a problem I continue to experience with all DMARDs & biologics. Unfortunately, it’s taking a second course of antibiotics to kick this infection which means I’ll be off of Xeljanz for almost three weeks while on antibiotics. This “interruption” in treatment is a recurring issue for me and makes it very difficult to find a viable treatment for the rheumatoid disease which is very active in all joints, chest wall, eyes, etc. I also choose to continue trying any treatment for the rheumatoid disease and to hope for improvement one day. Once I restart the Xeljanz, my rheumy will try a prophylactic antibiotic dose once per week in an attempt to prevent the recurring infections.
good luck Jeanie. lmk how you’re doing.
I am also one an antibiotic once daily because I have frequent bladder infections. My GP order a blood test to determine whether or not the antibiotic would raise the level of Methotrexate in my bloodstream. I developed a bit of a rash from the Xeljanz, but it seems to be clearing up. I can’t say the Xeljanz is doing miracles, but I am certainly doing better than on the methotrexate alone.
Kelly,
So sorry to hear this about your response to the new med. I was SO hoping to hear a positive report from you. I guess, we all just have to find “something” that makes us feel the “best” we can with what we have. I met with Dr. C last week. No real change since upping the MTX dosage. Actually, I felt worse. More tired, achey and swollen. So what did he do? Upped the MTX some more. Not sure exactly what 8 CC of 50 ml solution is but all I know is my system doesn’t like it at all. I am ready to quit and just go back to the basics at this point.
Thanks for all you do!
Amy
I had to stop cimzia after getting 3 sinus infections in 3 months. Now 3 weeks using Xeljanz. I am responding gradually. Better energy, much less pain in hands, feet & shoulders as the days go by. My Doctor says it wil continue to improve even more. I am so thankful to be responding. It is so hard to make changes in medications & wade into the unknown. The fight wears one out & it never stops.
While I think it is really awful the xeljanz didn’t work for you, I am so glad I am not alone. The last time I saw my rheumy, maybe a month ago, I had such severe hip pain I could barely walk when I got home or the next day, but doctor Insisted i was doing better since I had little swelling (I rarely have swelling). He took me off of it as I was having some neurological symptoms and I was only sleeping about 3 hours a night since starting the med (why he wouldn’t believe that is a listed side effect in the package insert??? But since none of his other pts experienced it). I also have a severe eczema rash that xeljanz completely cleared up but since being off it the rash is now back. At any rate, at least I don’t have to feel like a big faker that the med works for everyone else but me (that’s a pretty sad statement when you feel you doctor doesn’t believe you), but I think my presentation is different from a lot of people, I have some normal days, some horrible days and medication doesn’t seem to make much of a difference either way.
my ra doctor wants me to start xeljanz but the copay is around eight hundred a month and the side effects are so scary not sure what to do
RoseMarie why is the copay so high? I’m getting Xeljanz for 6.00copay Medicare approved me ..I have SilverScript ddrug plan.have you tried getting the free copay card from your doctors office.
Thank you for mentioning this. My RA doc had been wanting me to use Orencia and after taking Arava which if she had paid attention to my allergies would never have had me even take this. Yes I had horrible side effects and am allergic to sulfa drugs. This new med has me wondering if I should mention it to her and hope for some improvement. I did not know it was Medicare and Silverscript approved. I have both. Might be worth a try on the Xeljanez(sp?) I was on Humira for almost a year and did not notice mnay good things happening.
I’m doing great on it
Kelly, so sorry the Xeljanz did not help you.
rawarrior you’re not alone – I think Xeljanz is failing me too! How long were you on it? I’m around 6 weeks now – maybe too soon to give up
I am so disappointed that Xeljanz was not effective in relieving your RA. I may begin treatment with it within the next month so was hoping for a more optimistic report.
I had been on ACTEMRA for two years with some success. Earlier this year though I had the “rare” side effect of Actemra of a perforated colon and associated sepsis. Seventeen days in a hospital after emergency surgery with an ostomy and no RA treatment other than fairly low dose Medrol – I was so hoping for a different report. All of the other options have been exhausted to me – either no efficacy or side effects.
Keep us posted!
Barbara,
I’m sorry to hear about your complication of colon rupture. That is actually my biggest fear about actemra. I hadn’t heard from anyone this happened to, so thanks for sharing your story.
I also wanted to mention to you that xeljanz also lists GI/colon rupture or perforation as a possible complication. From what I have read, and heard from my rheumatologist, this is a problem not generally seen with other RA drugsm mostly the two newest ones, Actemra and Xeljanz. Just wanted to be sure you were aware of this so you can discuss with you doctor.
Xeljanz has not worked for me neither. I have been on it for 4.5 months without any improvement…just getting worse. Next will be remicade for me. This will be biologic number 9 so I am not getting my hopes up…but hope it helps some. I know those of you out there in my situation know exactly what I mean. Good luck to those of you who are just trying it, and so glad its working for those it is!
Just want to say, to those that haven’t found a effective med, that it can be worth it to keep trying new ones, if your situation (and insurance) allow it. I went through MTX, four different anti-TNFs, and Orencia with little to no results. Actemra turned out to be my magic bullet – been on it not quite two years, with near-complete remission and no noticeable side effects. Bloody amazing – the stuff worked almost as well for me as it does for those mythical patients in the stupid direct-to-patient ads.
Sure will be nice if they come up with a better way of figuring out who to put on what med then the “let’s put you on this stuff for six months and see what happens” approach, though.
I began my Xeljanz treatment about two weeks ago.
I had stopped taking MTX but have been on 10 mg prednisone for years.
The only med that worked for me was Rituxan but I nearly died during an infusion so go figure I am back at the starting point with a new med.
Tried nearly everything so far with serious infusion reactions and injection site reactions, stomach irritation, upper resp infections…you name it I have had it.
I will let you know as I wean off of the prednisone if the Xeljanz works. I am hopeful as I am only 31 years old and going at this for 7 years. I try to stay positive, but I am losing steam.
I just started Xeljanz 5 Mg.2xs a day.I’m in my 2nd.week.No side effects to report.I do feel better more energy.not depressed any more.
but it’s still early. But I’m very optimistic.
Still have some pain.taking pain meds for that.
Cutting back on them to see if the Xeljanz really is working! Hopeful! I’m about to crumble in my spine from prednisone over 38 yrs.We must tackle that now focus on rebuilding bone mass if it’s attainable.
I’ve been on Xeljanz for 1 month and am very significantly better after failing 4 TNFs and unable to take Methotrexate. Fatigue, dry eyes and overall deep body ache have all disappeared. My point of origination is my feet and they have as yet not been improved but my Rheumo tells me the peak of the med is 16 weeks and is hopeful they will “come along.” I miraculously have not had any side effects (when i had many others from injection site reaction w/Humira, awful headache on Enbrel and prickly all over on Simponi)but haven’t yet had a lab done so keeping my fingers crossed. I’m sorry for all of you who were hopeful and Xeljanz hasn’t worked for you but for those still trying or considering, keep the faith, there are those of us out there who are benefiting greatly from Xeljanz. Good luck.
“It has been my own personal choice to try every opportunity available to increase function and get relief from pain, and I’ve made that choice with no regrets.”
Thank you for this, Kelly!
I have been on Xeljanz for 6 months. I love it! It has given me my body back (not fully, but more functional). My biggest problem is the constant sinus infections. So I am on Doxycycline to try to keep them away.
whoohoo. Glad to hear it’s helping you, Sandra.
I’ve been on Xeljanz for two months, after failing several DMARDS, including methotrexate. I have peripheral neuropathy as well as RA. (Methotrexate and leflunomide made the neuropathy much worse, to the point of drowning out the RA symptoms!) So far the Xeljanz is helping, although the initial very good effect seems to be wearing off somewhat (sigh). I’m taking the drug in combination with Doxycycline, which reduces the neuropathy symptoms. My rheumatologist doesn’t believe that Doxycyline can have any beneficial effect, so the Doxy prescription is from my primary care doctor. I’m wondering if anyone else is taking Doxycyline for RA or a related condition? I know it’s helping my condition, but I’m not trying to convince my rheumatologist any longer — he is adamant that the drug cannot influence RA or neuropathy!
Does anybody have any insight into probiotics and if they are safe to use w/DMARDS? I have a burpy, churning stomach and am on Xeljanz. I would like to try a probiotic and see if it helps settle things down but was told back when i was on Humira by an Infectious Disease Doc that in his opinion “no one with a compromised immune system should take a probiotic”, that it is essentially “introducing bacteria into the system”.
Thoughts anyone??
Great questions Jane.
Maybe if you try it, it’s a good idea to separate the times as far as possible to avoid more tummy trouble. I guess that would be probiotics at lunchtime since Xj might be taken with breakfast and dinner.
I do know several people on biologics who take probiotics – well known & trusted brands of course.
Is anyone on sulfasalazine for their ra. I was on mtx for almost
A year but got too sick on it. Tried calling my
Doctor but she never called me back. Attempted
To call her and left over 20 messages for her. Finally
Went to a new ra doctor who put me on
Sulfasalazine . Can anyone tell me if this ra drug
Worked for them. Thanks so much.
I’m on Sulfasalazine. I am fairly newly diagnosed and feeling dreadful. Worse since starting the meds. I’m not sure if it’s a coincidence that my RA is getting worse and the meds haven’t taken full effect, or if the sulfasalazine is causing me to feel so bad. I started having foot pain in September 2012. I started having other symetrical joint pain by Feb. and had a RF factor of 616 in April. I started seeing a rheumy in May and started sulfasalazine. I have my follow up appt early August. I’m anxious to hear what he has to say because I’m really struggling right now. I’m exhausted about 50% of the time and have major muscle weakness and have had more joints become affected.
Hi Lisa,
I just wanted to follow up my original response now that I’m further along in my treatment. At the 12 week mark of having started sulfasalazine, I was still pretty bad and didn’t feel that it was working. My doc agreed and we added Enbrel to my treatment. Enbrel takes several weeks to really kick in as well. But week 13 I started to notice some improvement, though I hadn’t started the Enbrel. By week 15 I was definitely experiencing improvement even though Enbrel may not have been kicking in yet. So, I beleive the sulfasalazine did eventually help, though it took the full 12 weeks and beyond to really make a difference. Now that I’ve been on Enbrel and sulfasalazine together for 10 weeks I’m feeling better than I have all year. I hope you’re getting some relief as well.
Been on Xeljanz for almost a month now with 10cc methotrexate…non-responder to all other biologics I’ve tried as well as plaquenil. I thought the Xeljanz was actually helping…for about 2 weeks, but then it stopped (can’t phrase that any better)…now pain, stiffness and fatigue are worse than ever…still taking it, and hoping that it is a fluke and I feel better again…but am thinking it just quit.
I developed severe RA December 2012 and diagnosed February 2013. First prescribed MTX and a month later Simponi biologic. Simponi had limited affect after 4 months while still using MTX.
RA doc in August 2013, prescribed Xeljean together with MTX.
Happy to report Xeljean began working almost immediately to relieve the inflammation. I can now say after two weeks of taking Xeljanz inflammtion is gone from feet and ankles. There’s relatively little pain in hands, feet and other joints.
Xeljean was a miracle drug for me.
Yesterday spoke to the Professor of Rheumatology who headed up the 3 phase trials of Xeljanz.
Australia has decided NOT to allow Xeljanz onto our market as they feel the adverse events have outweighed the product.
Very interesting for those of us looking at different biologics.
After 20+ weeks on Xeljanz and an initial huge response to therapy, i must concede I think it is just “stopping”; the initial boost really only lasted about 9 weeks after which i slowly began losing ground. So discouraging…. I am switching up NSAIDs in the hope of getting back/jump starting the Xeljanz again but bit by bit i appear to be declining (stiff and sore, fatigue again, gritty eyes) and am beginning to think toward that next med…. Has anyone else had this experience with Xeljanz?
I am having the same response as you are having. My rheumy put me on the med nearly 6 months ago, and told me to come back in 6 months. He did tell me to call if I had any problems. Either he wants to “fire” me , or he is concerned that we have a 3 1/2 hour drive to St. Louis. In any case, I see him in about 3 weeks, so we’ll see where we go from here.
hello. I would really appreciate hearing about your followup w/your Rheumy re Xeljanz…i am in the process of evaluating the efficacy myself, began with an amazing response 6 months ago and ongoing decline in response. I’ve just had the the new Vectra Test (Cresendo Bioscience)to measure different markers and am awaiting results as we reevaluate disease activity and treatment plan. The Vectra is something you might want to consider as well. Good luck with that appointment:)
Someone mentions be good if we could figure out which biologic helps which people without having to go through them all ! Am wondering what could be the factor/answer ?
1. Age diagnosed with RA ? (I was 33 yrs. but had signs of it at around 25 yrs)
2. Is your RA hereditary (in the family genes) ?
3. Other conditions with your RA ?
4. I am mild Asthma, low/hypo thyroid, little bit psoriasis
5. What do you believe may have given u RA ? Workplace chemicals (printing ink) farm chemicals (sheep dip etc.)anything else ?
If you have a biologic that works for you maybe there is a common denominator ?
So … my rheumy just prescribed xeljanz for me and was checking comments here to see any consensus or news about this med. I have a different story in some ways from what I’m reading on this site but so much in common! I have moderate to severe (and actually more on the severe activity side) RA and no RA med as yet has worked to calm my inflammation and disease progression (mtx, sulfasalazine, actemra, nsaids all fail) Because I’ve been so sick with the RA and my whole immune system dysfunction, new liver problems, so many joints going bad, etc etc I started digging into all my labs and tests with one of my primary docs who practices alternative med. In doing so we discovered I carry the mutant genes for MTHFR which affect my ability to use folic acid and hence is the reason methotraxate was so toxic to me. (search pubmed and you will find that reference). And that spark of understanding some genetic background to the med’s that might work we found also I have Janus Kinase genetic mutations, JAK2, so when my rheumy discovered that she switched me to xeljanz which works to suppress this pathway since mine is broken. I just started taking it so we’ll see if it works! Anybody else with RA taking this drug with the jak2 mutation??
How did u test for jak problems? Xeljanz affects jak3 not jak2 so unsure how this is connected.
HI Guys
Just like to seek any ones thoughts or experience im a little lost and jaded.. with the use of Xeljanz pill and Actemra Infusions..
I was on Actemra 800mg monthly for about a year now with a weekly dose of MTX and then my Rheumy then said why not give Xeljanz a go as its getting good results. I am now 3 months on these pills and well to be honest not finding it as good as the monthly Actemra infusion.
I was wondering if any one has had this experience or situation like this.
The actmera worked fine but sort of tapered of towards the end of each month the last week of the month was ok but annoying.. then Boom another infusion and all was good hehe.
The Xeljanz took a month or so to work but I have noticed at least a 30% increase in pain and over all tiredness.. mornings really suck.. and horrible neck pain etc, its steady but do not find it as good as the Actemra.
would appreciate if any one has experiences like this or can comment on this.
Thanks every one for all the posts they really make life with RA more positive..
Rgds
Rory
Hi Kelly, First of all I cannot tell you how much your website has helped me, thank you! I was diagnosed with RA four years ago and probably had this awful disease for years. I have tried Five medicines I am not going to list them as they are the usual ones Drs start you on. I did not handle Methotrexate at all long story short I ended up with Remicade which really helped me. My insurance no longer would cover, so I was in a real pickle. Thought my life was really over because Remicade helped me so much. My Rheumatologist had some xlganz samples. Well it changed my life I feel I am in remission I am able to do the things I enjoy again. Like walk and pick up a golf club again! It’s been almost a year and I pray every day this disease will not rear its ugly self again. I am grateful and so thankful.
Kelly
I am about to start Xeljanz. I can’t tolerate Methotrexate at all – so it is out of any considerations. Just got off of Arava -, but only after my inflammation went up to 90 pts. and I was suffering daily migraines.
I have followed you from the beginning, love you and all of your advice and philosophy. Wish me luck!!!
Sherry
Good luck Sherry. Bless you!
I have been on Xeljanz since 2016, I have tried many different medication, oral, infusions, and injections, they seem to work for about 4 yrs then inflammation and pain come back. I’ve had RD for 30 yrs ,, even though I go to Aqua water exercise and try to live a normal life, I’m finding my body is starting to let me down through the deformities in which I now have. Rheumatologist say that they treat patients differently now, more aggressive at the start. Maybe I wouldn’t have such bad debilitating deformities if I had been treated differently. Last January I had major back surgery, 3 weeks later I had a Heart Attack. I’ve had feet and hand operations,
But I still enjoy life. You have to keep going.